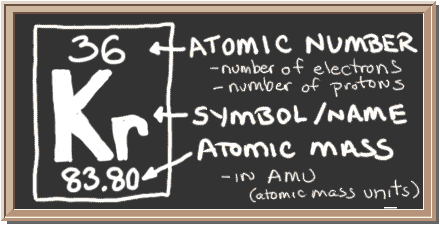
Check out the blackboard. That box on the left has all of the information you need to know about one element. It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table.
Now we're working with the fourth period/row in the table of elements. You may have an easy way to know the number of electrons in a neutral atom, but the placement of those electrons gets a little more complex. Let's take a look at the arrangements of electrons in the basic elements (left and right sides of the table) of period four and the more complex arrangements of the transition elements (in the middle of the row). If you think this is a little over your head, go back and look at the elements 1-18 that have organizations that are a little more simple.
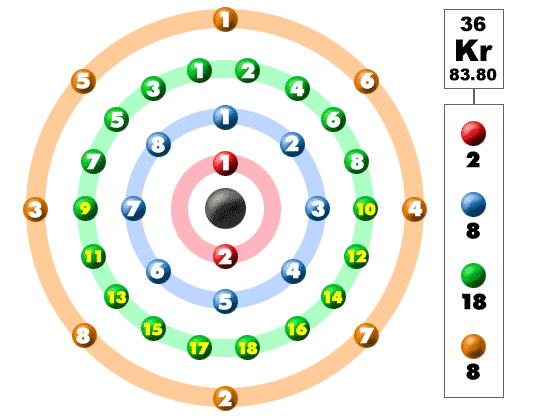
Electrons In The Shells
Take a look at the picture below. Each of those colored balls is an electron. In an atom, the electrons spin around the center, also called the nucleus. The electrons like to be in separate shells/orbitals. As you learn more about atomic structure, you will learn that the electrons don't stay in defined areas around the nucleus. They are found in clouds that can have different shapes that include spheres and dumbbell-like shapes. So remember when you look at our breakdown that the electrons aren't always in a nice neat order as shown here.Since krypton is in the far right row of the periodic table, its outermost shell is full with eight electrons. This is one of the happy elements and has an electron configuration of 2-8-18-8. The other inert gases including argon and xenon also have full outer shells with eight electrons.
The inert gases have a valence number of 0. They prefer no to make compounds with other elements and stick to themselves. Krypton is a bit of an exception. Some compounds have been made in labs with this noble gas (fluorine compounds). It's not an easy process, but it can be done. It's kind of like teaching you to memorize the lanthanide series of elements. Difficult, but possible.